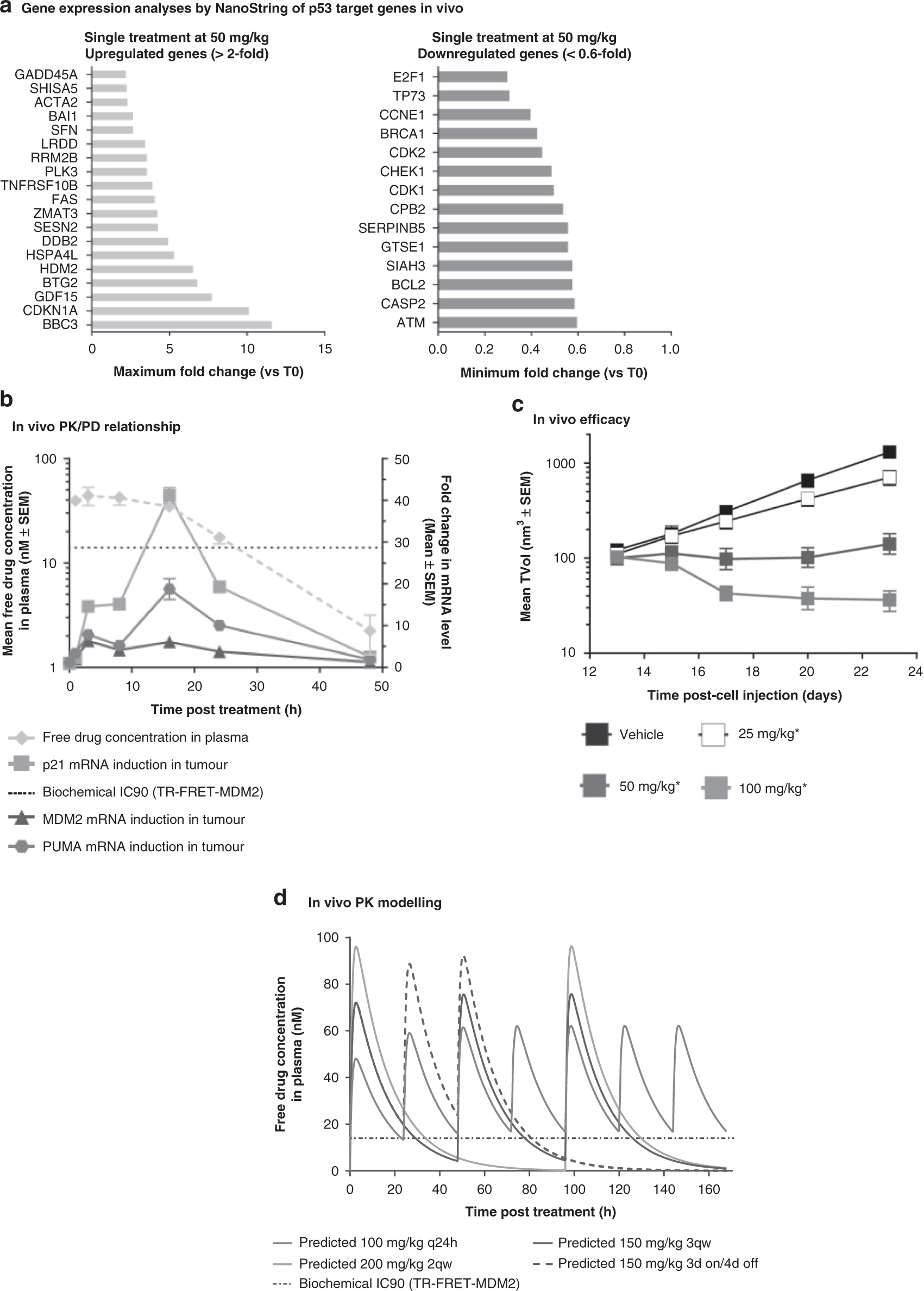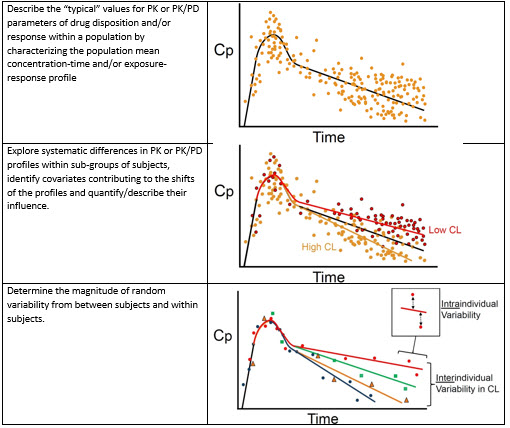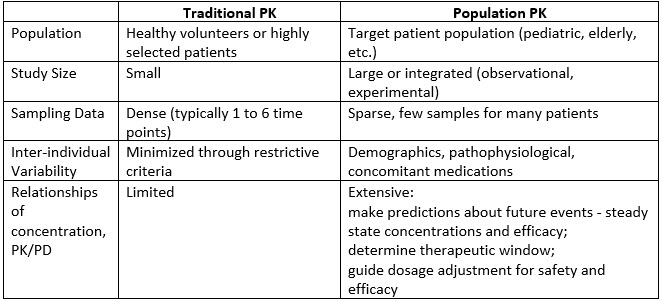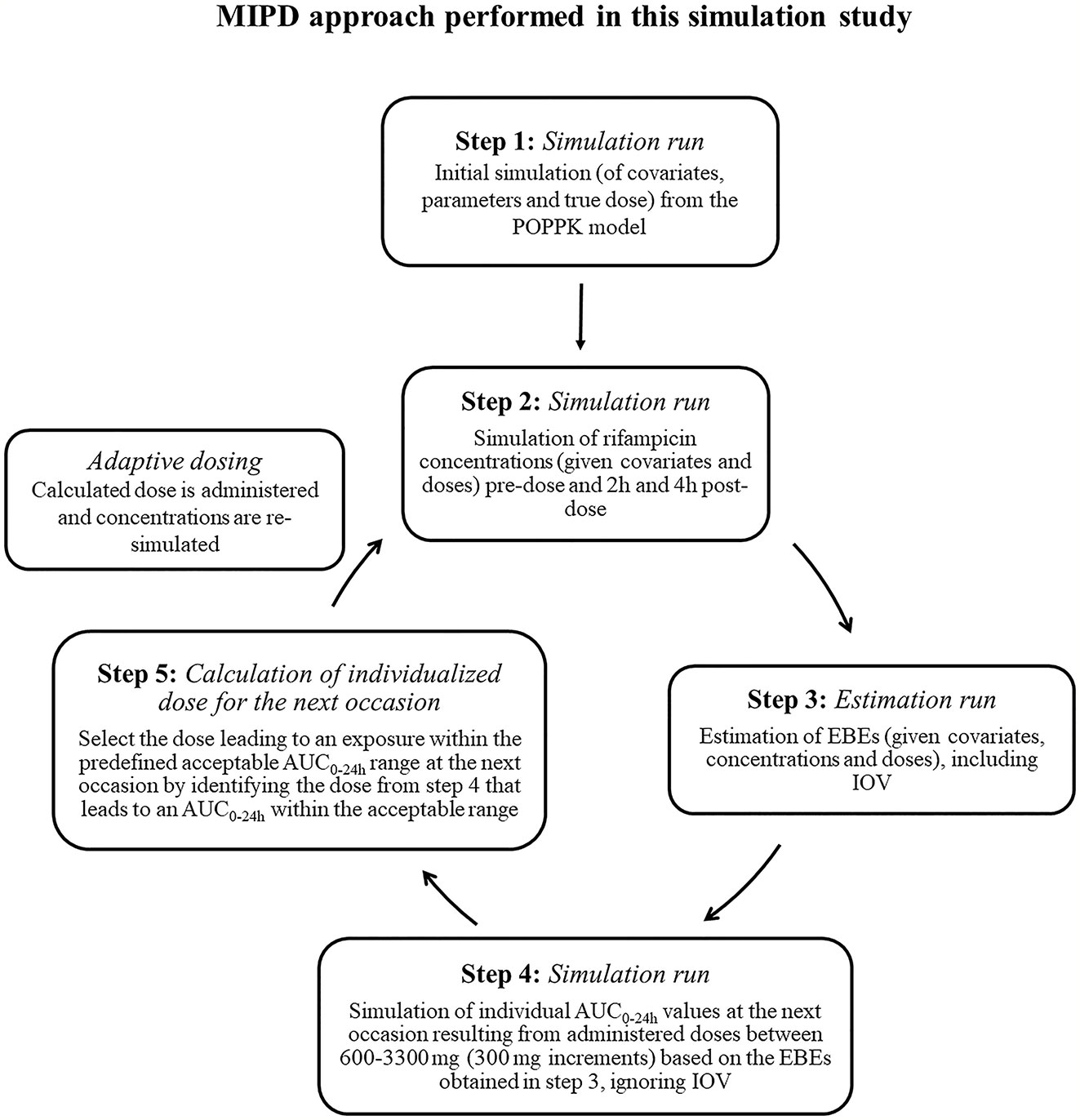
Frontiers | Individualized Dosing With High Inter-Occasion Variability Is Correctly Handled With Model-Informed Precision Dosing—Using Rifampicin as an Example
Session 1: An Introductory Workshop in Population PK/PD Data Analysis with NONMEM® (In Person) - School of Pharmacy and Pharmaceutical Sciences - University at Buffalo
Prior information for population pharmacokinetic and pharmacokinetic/pharmacodynamic analysis: overview and guidance with a focus on the NONMEM PRIOR subroutine | SpringerLink

Pharmacokinetics and population pharmacokinetics in pediatric oncology - Sassen - 2020 - Pediatric Blood & Cancer - Wiley Online Library

Fitusiran Population Pharmacokinetic and Pharmacodynamic (PopPK/PD) Modeling to Support Revised Dose, Dosing Regimens & Dose Mitigation Scheme - ISTH Congress Abstracts

Pharmaceutics | Free Full-Text | Matrix Effects of the Hydroethanolic Extract of Calyces of Physalis peruviana L. on Rutin Pharmacokinetics in Wistar Rats Using Population Modeling

A Novel Integrated Pharmacokinetic-Pharmacodynamic Model to Evaluate Combination Therapy and Determine In Vivo Synergism | Journal of Pharmacology and Experimental Therapeutics

Population Pharmacokinetics of an Anti-PD-1 Antibody Camrelizumab in Patients with Multiple tumor types and model informed dosing strategy | bioRxiv

Simulation results using PK parameters obtained from PopPK analysis... | Download Scientific Diagram

Combining “Bottom‐up” and “Top‐down” Approaches to Assess the Impact of Food and Gastric pH on Pictilisib (GDC‐0941) Pharmacokinetics - Lu - 2017 - CPT: Pharmacometrics & Systems Pharmacology - Wiley Online Library

A Survey of Population Pharmacokinetic Reports Submitted to the USFDA: An Analysis of Common Issues in NDA and BLA from 2012 to 2021 | SpringerLink

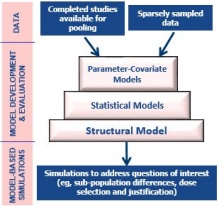
Establishing Best Practices and Guidance in Population Modeling: An Experience With an Internal Population Pharmacokinetic Analysis Guidance - Byon - 2013 - CPT: Pharmacometrics & Systems Pharmacology - Wiley Online Library

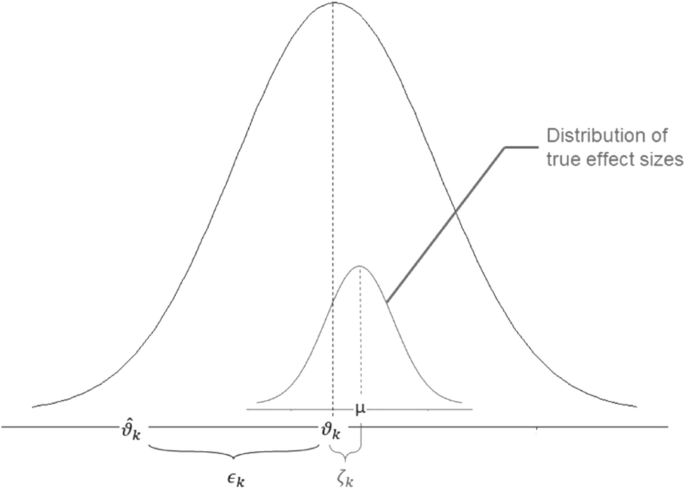
Population Pharmacokinetic Variables: THETA, ETA, EPSILON; and NONMEM for analysis - Pharmacoengineering